|
9/28/2023 0 Comments Can entropy decreaseFinally, substances with strong hydrogen bonds have lower values of S°, which reflects a more ordered structure. For example, compare the S° values for CH 3OH(l) and CH 3CH 2OH(l). Similarly, the absolute entropy of a substance tends to increase with increasing molecular complexity because the number of available microstates increases with molecular complexity. To find the entropy change of the system, we took the standard entropy changes of products minus reactants and got -262J/molK. Soft crystalline substances and those with larger atoms tend to have higher entropies because of increased molecular motion and disorder. begingroup We found the entropy change of the universe for the photosynthesis process to be negative. Removal of matter from a system can also decrease its entropy. According to the second law of thermodynamics, the total entropy of an isolated system remains constant. Differing from Planck's just foregoing principle, this one is explicitly in terms of entropy change. Entropy cannot be destroyed by any means, at any scale, and thus, entropy cannot overall decrease. In contrast, graphite, the softer, less rigid allotrope of carbon, has a higher S° due to more disorder in the crystal. there is only one way in which the entropy of a closed system can be decreased, and that is to transfer heat from the system. Entropy is generated everywhere and always (and thus overall increased), at any scale without exception (including life processes, open systems, micro-fluctuations, gravity, or entanglement). Among crystalline materials, those with the lowest entropies tend to be rigid crystals composed of small atoms linked by strong, highly directional bonds, such as diamond. The entropy of the cup of coffee falls (and the entropy of the surrounding air increases). Put the cup on a table and wait while it cools. 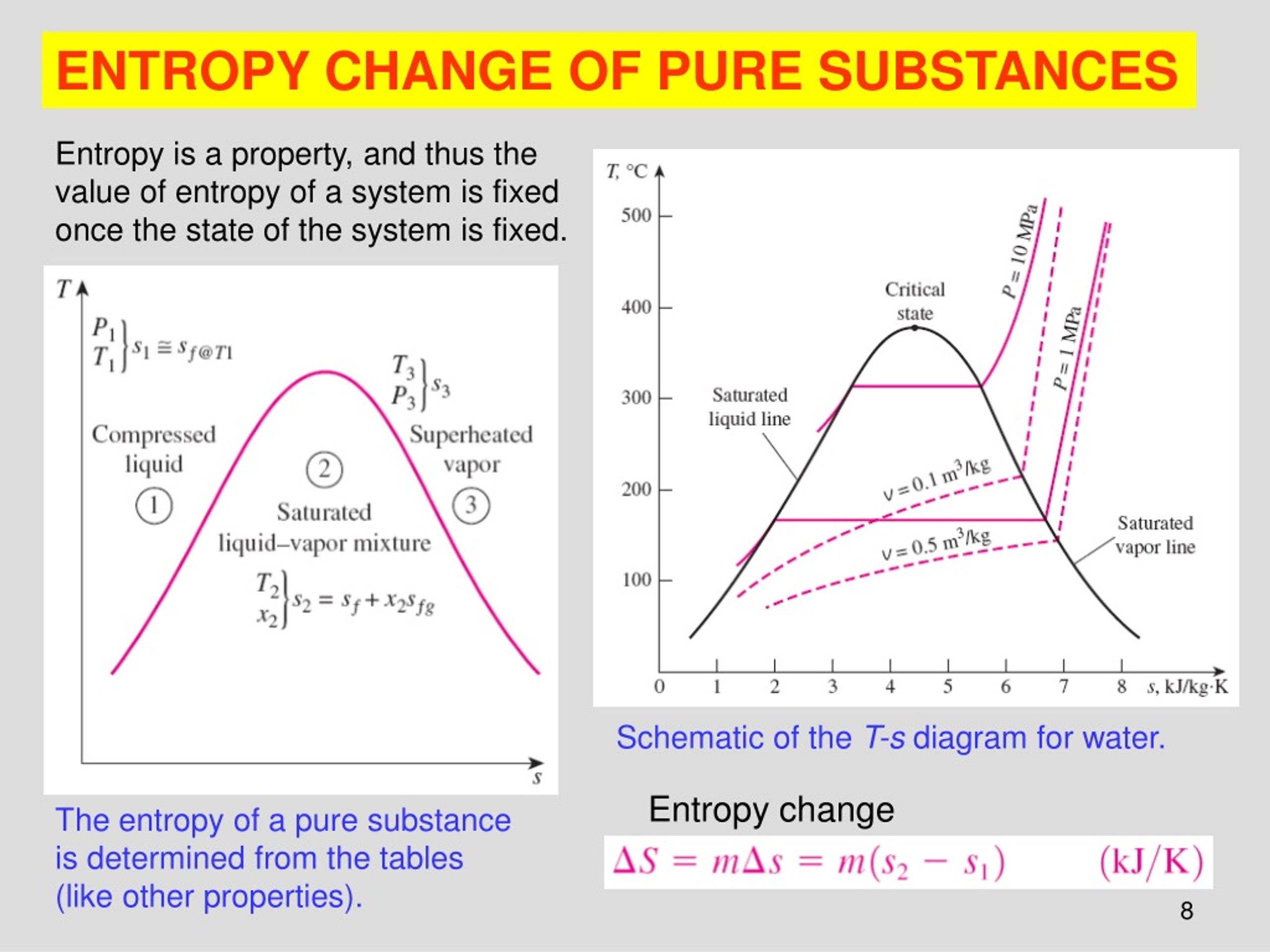 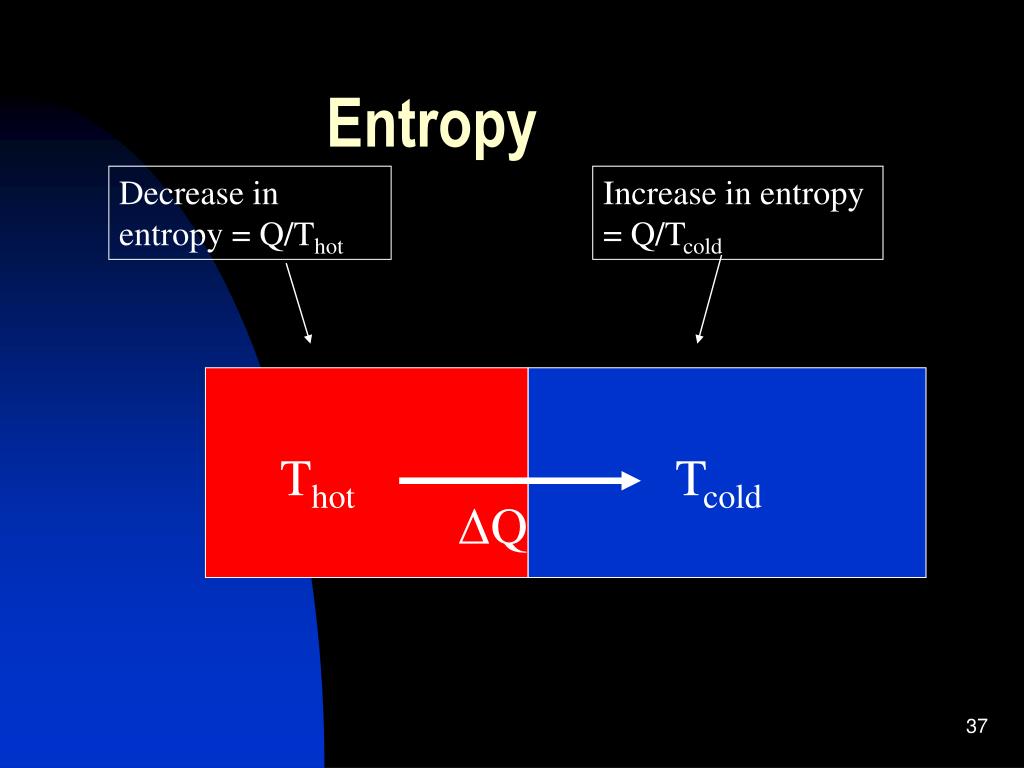
\( \newcommand\) also reveals that substances with similar molecular structures tend to have similar S° values. All you need is for heat to flow away from the local region.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
